 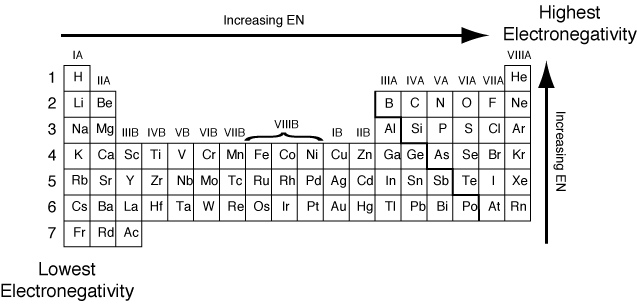
If the atoms that form a covalent bond are identical, as in \ce.Įxample 6.2. Furthermore, whereas ionic compounds are good conductors of electricity when dissolved in water, most covalent compounds are insoluble in water since they are electrically neutral, they are poor conductors of electricity in any state. It is measured using the Pauling scale fluorine is highest at 4.0 on the scale, the most electronegative element, whilst francium is the lowest at 0.7 and is. Electronegativity is a measure of how much an atom attracts electrons.For instance, a more electronegative atom will be easily reduced, while a less electronegative atom will be easily oxidized. In fact, many covalent compounds are liquids or gases at room temperature, and, in their solid states, they are typically much softer than ionic solids. Because the attraction between molecules, which are electrically neutral, is weaker than that between electrically charged ions, covalent compounds generally have much lower melting and boiling points than ionic compounds. Provides a complete periodic table of electronegativity values. 
Define electronegativity and assess the polarity of covalent bondsĬompounds that contain covalent bonds exhibit different physical properties than ionic compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
